Back to Basics – Variations – Types & Timelines
Here is the second installment of our Back to Basics series: VARIATIONS
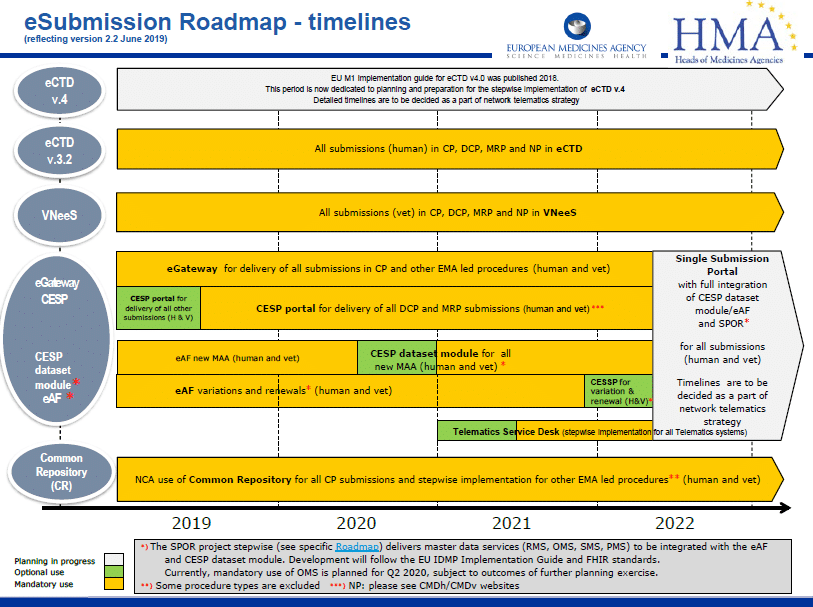
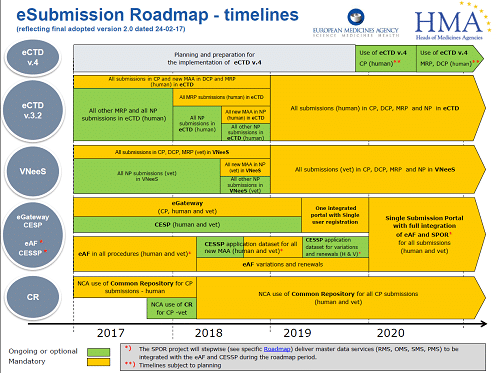
So, let’s start with a summary of the different types of variations & their timelines.
Variations are
- changes made to the dossier of an authorised medicinal product after its initial registration
- They may concern administrative changes, Quality changes, Safety/Efficacy changes or Vigilance changes
- Variations can be categorised as Type IAIN, Type IA, Type IB (seen or foreseen) or Type II whether they are within a Centralised, Decentralised (DCP) or National Procedure (NP)
- Variations can either be a single or grouped together.
- There is also a mechanism to submit variations by a work sharing procedure.
Procedures around variations are currently governed and harmonised throughout the EU by Commission Regulation (EC) No 1234/2008 of 24 of November 2008 (“the Variations Regulation”) which has subsequently been amended by Regulation (EU) 712/2012.
| Variation Types | Definition | Timelines |
| Human Medicines Regulations | ||
| Type IAIN
|
A minor variation which has only minimal/or no impact on the quality, safety or efficacy of the medicinal product. The competent authority should be notified immediately after implementation.
‘Immediately’ is not defined in EU guidance but is generally considered to be within 2 or so weeks of implementation* of the change.
*For quality changes, ‘implementation’ is when the company makes the change in its own quality system. For product information, it is when the company internally approves the revised product information. The revised product information will then be used in the next packaging run. e.g. to introduce a PSMF or change company address |
|
| Type IA | A minor variation which has only minimal/or no impact on the quality, safety or efficacy of the medicinal product.
Where all required data conditions are met such variations do not require any prior approval, but must be notified by the MAH within 12 months following implementation in so called ‘annual reports’ or may be submitted earlier should this facilitate dossier life-cycle maintenance or when necessary. This is known as the ‘Do and tell’ procedure. e.g. updated CEP from an already approved manufacturer |
|
| Type IB | Variations which are neither a minor variation of Type IA nor a major variation of Type II nor an extension are classified as Type IB variation by default.
They are further classified as being either foreseen or unforeseen (consult variation guidance documents referenced in section 4, below, for further information) e.g. change of product name for nationally authorised products |
|
| Type II | Type II variations are classified as major variations. They arise from a change that does not meet the requirements of a Type I variation, but is not so complex as to lead to a new Marketing Authorisation application. There are two classifications of Type II variations:
(1) Type II Complex (2) Type II Urgent Safety Type II variations require a formal approval e.g. addition of a new therapeutic indication |
|
Grouping:
Where the same variation(s) affect one or more marketing authorisation(s) from the same holder, the marketing authorisation holder may choose to submit these variations as one application.
In most instances it is possible to group type IA variations regardless of content.
Some competent authorities may not allow grouping for national variations.
In specified cases it is possible to group type IA and type IB/type II variations (consult acceptable and non-acceptable grouping guidance document referenced below, for further information. In these cases the changes will tend to be consequential and the procedure type defaults to the highest recorded procedure.
The website of the relevant competent authority should be referred to, to confirm the possibility of grouping.
For grouping that includes type IB and type II procedures and falls outside the scope of acceptable and non-acceptable grouping guidance documents, consent for the grouping should be sought in advance from the competent authority to avoid rejection.
Worksharing:
Article 20 of the Commission Regulation (EC) No 1234/2008 allows for the possibility for the Marketing Authorisation Holder to submit the same type IB or type II variation or the same group of variations affecting more than one marketing authorisation from the same Marketing Authorisation Holder in one application.
A worksharing procedure allows for one authority to be chosen amongst the competent authorities of the member states to examine the variation on behalf of the other concerned authorities. In case a grouped application is applied this may also contain consequential IA changes.
Line extensions are excluded from worksharing.
Further to above, Applicants may wish to pay particular attention to the following CMDh documents:
- EC Guidelines
- Best Practice Guides for Submission and Processing of Variations
- Chapter 3 – CMDh Best Practice Guide for the processing of Type IA minor variations (notifications) in the Mutual Recognition Procedure
- Chapter 4 : CMDh BPG for the processing of Type IB Minor Variations (Notifications) in the Mutual Recognition Procedure
- Chapter 5: CMDh BPG for the handling of Type II Variations in the Mutual Recognition Procedure
- Examples of acceptable and not acceptable groupings
- Questions & Answers on variations
- Chapter 8 – CMDh Best Practice Guide on CMDh recommendations on unforeseen variations
- Requirements on submissions for Variations and Renewals within MRP and National Procedures
Feel free to contact us here at ERA to assist you with all things regulatory in Ireland, UK and across the EU.
We take the pain out of regulatory so that you can take your medicine to the next level.
Written by
Marian Winder